
| Taxa: |
| Order: |
| Family: |
| Reptilia |
| Squamata |
| Colubridae |
| NatureServe Global Rank: |
| NatureServe State (NC) Rank: |
| G5 |
| S3 |
| Federal Status: |
| NC State Status: |
| --- |
| --- |
| Land Unit |
| US Fish & Wildlife Service |
| US Forest Service |
| US National Park Service |
| US Department of Defense |
| NC State Parks |
| NC University System |
| NC Wildlife Res. Com. |
| NC Forest Service |
| NC Div. of Coastal Mgmt. |
| Local Governments |
| Non-Governmental Org. |
| Other Public Lands |
| Private Lands |
| GAP Status 1-2 |
| All Protected Lands |
| Statewide |
| Hectares |
| 74.43 |
| 5,452.02 |
| 47,199.60 |
| 2,009.52 |
| 453.42 |
| 0.00 |
| 19,062.45 |
| 39.15 |
| 0.00 |
| 184.77 |
| 776.70 |
| 64.98 |
| 275,561.73 |
| 4,387.77 |
| 74,747.88 |
| 350,878.77 |
| Acres |
| 183.92 |
| 13,472.23 |
| 116,632.73 |
| 4,965.63 |
| 1,120.43 |
| 0.00 |
| 47,104.33 |
| 96.74 |
| 0.00 |
| 456.58 |
| 1,919.27 |
| 160.57 |
| 680,927.73 |
| 10,842.41 |
| 184,706.00 |
| 867,040.15 |
| % of Dist. on |
| Prot. Lands |
| < 0.1 % |
| 7.3 % |
| 63.1 % |
| 2.7 % |
| 0.6 % |
| 0.0 % |
| 24.8 % |
| < 0.1 % |
| 0.0 % |
| 1.0 % |
| 1.0 % |
| < 0.1 % |
| 0.0 % |
| 5.9 % |
| ----- |
| ----- |
| % of Dist. on |
| All Lands |
| < 0.1 % |
| 1.6 % |
| 13.5 % |
| 0.6 % |
| 0.1 % |
| 0.0 % |
| 5.4 % |
| < 0.1 % |
| 0.0 % |
| < 0.1 % |
| 0.2 % |
| < 0.1 % |
| 78.5 % |
| 1.3 % |
| ----- |
| ----- |
|
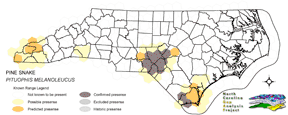
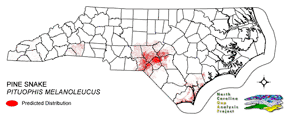
NATURE SERVE GLOBAL HABITAT COMMENTS: Lowlands to mountains; desert, prairie, brushland, woodland, open coniferous forest, farmland, marshes. Eastern populations occupy sandy pine woods and dry shaly ridges; midwestern population inhabit prairies; western and Mexican populations occur from coastal grasslands and forests through deserts into montane forests (Sweet and Parker 1990). Terrestrial, fossorial, and arboreal. Remains underground in cold weather and during hot midday period in summer (Fitch 1956, Zappalorti et al. 1983, Burger et al. 1988); may occupy mammal burrow (Schroder 1950, Fitch 1958) or dig own burrow (pine snakes and bullsnakes, aided by a pointed snout and enlarged rostral scale, are accomplished burrowers in loose soil [Franz 1992]; Carpenter [1982] estimated that burrowing PITUOPHIS could move up to 3400 cubic cm of soil in an hour). Eggs are deposited in burrows excavated by the female in loose soil (Moore 1893; Zappalorti et al. 1983; Burger and Zappalorti 1986, 1991), spaces beneath large rocks or logs, or possibly small mammal burrows (Ernst and Barbour 1989, Franz 1992). Good quality pine snake habitat appears to be characterized by xeric, pine-dominated or pine-oak (50 to 80% pine) woodland with an open, low understory established on sandy soils. Longleaf pine sandhills appear to represent critical habitat over much of the southeastern U.S. Pine snakes also require forest openings, with level, well-drained sandy soils and little shrub cover, as nesting and hibernation sites. New Jersey: habitat is restricted to the open Pine Barrens of the southern portion of the state (Zappalorti et al. 1983). Ten radio-telemetered individuals tended to be found in dense vegetation that provided shade and moist, cool ground cover (Burger and Zappalorti 1988). Snakes were inactive 41% of the time, basked 36%, were moving 20%, and nested 3%. Selected habitat was primarily (93% of locations) pitch pine-scrub oak (QUERCUS MARILANDICA) uplands (50-80% pine); Atlantic white cedar (CHAMAECYPARIS THYOIDES) swamps and lowland pine areas were avoided. Snakes spent roughly equal amounts of time in human-altered and undisturbed habitats, with no major seasonal habitat shifts other than at nesting and hibernation. Early successional habitats in sites that had been previously disturbed by human activity appeared to be of crucial importance in both foraging and nesting (Zappalorti and Burger 1985). Habitat selection of radio-telemetered pine snakes differed appreciably from the apparent habitat selection of hand-captured or randomly encountered snakes (Burger and Zappalorti 1988). Thermoregulation appeared to be important in habitat selection. Snakes were inactive at very high and very low temperatures, when they remained under wet leaves or inside hollow logs. On windy days they sought cover in blueberry (VACCINIUM spp.) patches and basked partially covered by vegetation, possibly to avoid dehydration (Burger and Zappalorti 1989). Hibernacula and summer dens in New Jersey usually were beside old fallen logs, with entrance tunnels following decaying roots into soil. Snakes in hibernacula were at depths of 50-110 cm. Hibernacula were characterized by lower surface and soil temperature, higher surrounding leaf cover (mean = 55%), were closer to trees (mean = 130 cm), and had more trees within 10 m (mean = 138) than random points in clearings. In general, hibernacula were relatively more protected sites than random points and were less vulnerable to daily temperature fluctuations. Hibernacula appeared to have been excavated by the snakes themselves. Typically they had an average of eight side chambers and 642 cm of tunnels. Except for hatchlings, most snakes in hibernacula were located in individual chambers off the main tunnel and not in groups. All snakes were found at depths of 50 to 111 cm (mean = 79 cm). All hibernacula were within 100 m of nesting sites and most were only 10 to 15 m from nest chambers. See Burger et al. (1988) for further details. In New Jersey, eggs were deposited in nests as much as 25-30 cm deep. Nest sites often were re-used in subsequent years (Burger et al. 1988; Burger and Zappalorti 1991, 1992). Excavation took 2-4 days to complete and not all females that excavate nests were are gravid (Burger and Zappalorti 1991). Of 93 nest sites studied by Burger and Zappalorti (1986, 1991) all were located in large clearings with less than 10% tree cover in pitch pine-scrub oak xeric uplands. Nest sites were in soft sand in open, unvegetated sections of clearings, either recently cleared or burned areas with exposed sand. Clearings used for nesting averaged 166 m long and 79.2 m wide with slopes less than 14 degrees. Ground cover averaged less than 5% over the entire nest, less over the mouth and egg chamber. Nests were excavated in soft packed sand with little or no humus. Snakes appeared to select a particular range of sand hardness that provided ease of digging but was firm enough to avoid burrow collapse. Soil moisture may have been important in site selection (Burger and Zappalorti 1986, 1991). Nests ranged between 90-305 cm (mean = 187.6 cm) in length, averaged about 14 cm wide, and eggs were found at an average depth of about 21 cm below the surface. All nests were located in clearings or along roads or railroad beds. Clearings were usually man-made (only 1 or 20 clearings appeared to be naturally created by fire). Burger and Zappalorti (1986) suggested that man-made clearings were crucial to nesting ecology in the New Jersey Pine Barrens and may have improved nesting habitat for this population. North Carolina: longleaf pine (PINUS PALUSTRIS) sandhills (Brimley 1944). South Carolina: Gibbons et al. (1991) described the habitat as sandy old fields and turkey oak (QUERCUS LAEVIS)-pine forests. Louisiana: both P. M. LODINGI and P. M. RUTHVENI are restricted to longleaf pine forests and second growth longleaf pine- blackjack oak (QUERCUS MARILANDICA) associations (Fugler 1955, Walker 1965). Both P. M. LODINGI and P. M. RUTHVENI appear to use the burrows of pocket gophers, gopher tortoises, and armadillos (Dundee and Rossman 1989). Texas: Mitchell and Tinkle (1960) reported observing P. M. RUTHVENI foraging in a seasonally dry, acid bog, where the vegetation consisted of loblolly pine (PINUS TAEDA) and shortleaf pine (PINUS ECHINATA) with a dense sweetbay (MAGNOLIA VIRGINIANA) and cyrilla (CYRILLA RACEMIFLORA) understory. Florida: subspecies MUGITUS is found in xeric sites, occurring primarily in longleaf pine-turkey oak woodlands, but also in sand pine (PINUS CLAUSA) scrub, pine flatwoods on well-drained soils, and old fields on former sandhill sites (Franz 1992). Pine snakes seek out open habitats surrounded by wetlands during drought (Franz 1992). Franz (1992) also reported Florida pine snakes to actively seek burrows of gopher tortoises (GOPHERUS POLYPHEMUS) as shelter. NATURE SERVE STATE HABITAT COMMENTS: On Coastal Plain these snakes are found in relatively open forests of longleaf pine, wiregrass and turkey oaks; and also in dry piney flatwoods. Little is known of mountainous habitats. REPRODUCTIVE HABITAT COMMENTS: Eggs laid in open areas exposed to the sun, in burrows or cavities underground (B83NUS01NA, A86BUR02NA), or under large rocks or logs (B79BEH01NA). |
| Code | Name | Description | NC Natural Heritage Program Equivalent |
| 67 | Wet Longleaf or Slash Pine Savanna | Wet flatwoods and pine savannas, typically dominated by longleaf pines, but slash or pond pines may be the dominant pines. | Wet Pine Flatwoods |
| 97 | Mesic Longleaf Pine | Longleaf pine woodlands without a major scrub oak component. Slash or loblolly pines may be present as well. | Mesic Pine Flatwoods |
| 42 | Xeric Longleaf Pine | Sandhills including a range of longleaf pine density from predominantly wiregrass, scrub oak dominated to true longleaf pine woodland. This does not include mesic or saturated flatwood types. | Xeric Sandhill Scrub, Pine/Scrub Oak Sandhill, Coastal Fringe Sandhill |
| 46 | Xeric Oak - Pine Forests | Mixed forest dominated by yellow pines with white or northern red oaks co-dominating. | Pine Oak Heath |
| 232 | Xeric Pine-Hardwood Woodlands and Forests | Mixed forest dominated by yellow pines with drier oaks including southern red, post, and chestnut oaks. | Dry Oak Hickory Forest |
| 220 | Piedmont Xeric Pine Forests | Dry to xeric pine forests dominated by Virginia pine, shortleaf pine or Eastern Red Cedar. | Pine Oak Heath |
| 226 | Piedmont Xeric Woodlands | Generally post and blackjack oak dominated woodlands. White ash and pignut hickory can be found in combination with Eastern red cedar on glades. | Xeric Hardpan Forest |
| 528 | Appalachian Xeric Pine Forest | Pine forests and woodlands on xeric sites. A variety of pines, including Virginia, Shortleaf, Eastern White Pine, Table Mountain and Pitch pine. Often small areas of dense pine within a matrix of Xeric Oak-Pine Forests. | Pine Oak Heath |
|
Carpenter, C. C. 1982. The bullsnake as an excavator. Journal of Herpetology 16:394-401.
Sweet, S. S., and W. S. Parker. 1990. PITUOPHIS MELANOLEUCUS. Cat. Am. Amph. Rept. 474.1-474.8. Eichholz, M. W., and W. D. Koenig. 1992. Gopher snake attraction to birds' nests. Southwest. Nat. 37:293-298. Franz, R. 1992. Florida pine snake PITUOPHIS MELANOLEUCUS MUGITUS Barbour. Pages 254-258 in P. E. Moler, editor. Rare and endangered biota of Florida. Vol. III. Amphibians and reptiles. Univ. Press of Florida. Moore, J. M. 1893. The eggs of PITUOPHIS MELANOLEUCUS. Am. Nat. 27:878-85. Diller, L. V., and R. L. Wallace. 1996. Comparative ecology of two snake species (CROTALUS VIRIDIS and PITUOPHIS MELANOLEUCUS) in southwestern Idaho. Herpetologica 52:343-360. Conant, R. and J. T. Collins. 1991. A field guide to reptiles and amphibians:eastern and central North America. Third edition. Houghton Mifflin Co., Boston, Massachusetts. 450 pp. Gibbons, J. W., and R. D. Semlitsch. 1991. Guide to the reptiles and amphibians of the Savannah River Site. Univ. of Georgia Press, Athens. xii + 131 pp. Reichling, S. B. 1995. The taxonomic status of the Louisiana pine snake (PITUOPHIS MELANOLEUCUS RUTHVENI) and its relevance to the evolutionary species concept. Journal of Herpetology 29:186-198. Schroder, R. C. 1950. Hibernation of blue racers and bull snakes in western Illinois. Natur. Hist. Misc. 75:1-2. Burger, J., and R. T. Zappalorti. 1989. Habitat use by pine snakes (PITUOPHIS MELANOLEUCUS) in the New Jersey Pine Barrens:individual and sexual variation. J. Herpetol. 23:68-73. Fugler, C. M. 1955. New locality records for the Louisiana pine snake, PITUOPHIS CATENIFER RUTHVENI Stull. Herpetologica 11:24. Fitch, H. S. 1956. Temperature responses in free-living amphibians and reptile in northeastern Kansas. University of Kansas Publication Museum of Natural History 8:417-76. Fitch, H. S. 1958. Home ranges, territories, and seasonal movement of vertebrates of the Natural History Reservation. University of Kansas Publication Museum of Natural History 11:63-326. Mitchell, R. W. and D. W. Tinkle. 1960. Another Texas record for the Louisiana pine snake, PITUOPHIS MELANOLEUCUS RUTHVENI Stull. Herpetologica 16:143-4. Zappalorti, R. T., E. W. Johnson, and Z. Leszczynski. 1983. The ecology of the northern pine snake, PITUOPHIS MELANOLEUCUS (Daudin) (Reptilia, Serpentes, Colubridae) in southern New Jersey, with special notes on habitat and nesting behavior. Bulletin of t Fitch, H. S. 1970. Reproductive cycles of lizards and snakes. Univ. Kansas Museum Natural History Miscellaneous Publication 52:1-247. Walker, J. M. 1965. Notes on two rare Louisiana serpents. Herpetologica 21:159-60. Mount, R. H. 1975. The Reptiles and Amphibians of Alabama. Auburn University Agricultural Experiment Station, Auburn, Alabama. vii + 347 pp. Behler, J. L., and F. W. King. 1979. The Audubon Society field guide to North American reptiles and amphibians. Alfred A. Knopf, New York. 719 pp. Parker, W. S., and W. S. Brown. 1980. Comparative ecology of two colubrid snakes, Masticophis t. taeniatus and Pituophis melanoleucus deserticola, in northern Utah. Milwaukee Pub. Mus. Pub. Biol. Geol. 7. 104 pp. Zappalorti, R. T., and J. Burger. 1985. On the importance of disturbed sites to habitat selection by pine snakes in the Pine Barrens of New Jersey. Environmental Conservation 12(4):358-361. Burger, J., and R. T. Zappalorti. 1986. Nest site selection by pine snakes, PITUOPHIS MELANOLEUCUS, in the NewJersey Pine Barrens. Copeia 1986:116-121. Nussbaum, R. A., and E. D. Brodie, Jr. and R. M. Storm. 1983. Amphibians and Reptiles of the Pacific Northwest. University Press of Idaho. 332 pp. Knight, J. L. 1986. Variation in snout morphology in the North American snake PITUOPHIS MELANOLEUCUS (Serpentes:Colubridae). J. Herpetol. 20:77-79. Tennant, A. 1984. The Snakes of Texas. Texas Monthly Press, Austin, Texas. 561 pp. Stebbins, R. C. 1985. A Field Guide to Western Reptiles and Amphibians. Second Edition. Houghton Mifflin Company, Boston, Massachusetts. xiv + 336 pp. Green, N. B., and T. K. Pauley. 1987. Amphibians and reptiles in West Virginia. University of Pittsburg Press, Pittsburg, Pennsylvania. xi + 241 pp. Burger, J., and R. T. Zappalorti. 1991. Nesting behavior of pine snakes (PITUOPHIS M. MELANOLEUCUS) in the New Jersey Pine Barrens. J. Herpetol. 25:152-160. Collins, J. T. 1991. Viewpoint:a new taxonomic arrangement for some North American amphibians and reptiles. SSAR Herpetol. Review 22:42-43. Dundee, H. A., and D. A. Rossman. 1989. The amphibians and reptiles of Louisiana. Louisiana State Univ. Press, Baton Rouge. Ernst, C. H., and R. W. Barbour. 1989. Turtles of the world. Smithsonian Institution Press, Washington, D.C. xii + 313 pp. Collins, J. T. 1990. Standard common and current scientific names for North American amphibians and reptiles. SSAR Herpetol. Circular No. 19. 41 pp. Burger, J., and R. T. Zappalorti. 1988. Habitat use in free-ranging pine snakes, PITUOPHIS MELANOLEUCUS, in New Jersey Pine Barrens. Herpetologica 44:48-55. Burger, J., et al. 1988. Hibernacula and summer den sites of pine snakes (PITUOPHIS MELANOLEUCUS) in the New Jersey Pine Barrens. J. Herpetol. 22:425-433. Diller, L. V., and D. R. Johnson. 1988. Food habits, consumption rates, and predation rates of western rattlesnakes and gopher snakes in southwestern Idaho. Herpetologica 44:228-233. Burger, J., and R. T. Zappalorti. 1992. Philopatry and nesting phenology of pine snakes PITUOPHIS MELANOLEUCUS in the New Jersey Pine Barrens. Behav. Ecol. Sociobiol. 30:331-336. Burger, J., et al. 1992. Subterranean predation on pine snakes (PITUOPHIS MELANOLEUCUS). J. Herpetol. 26:259-263. |
For more information please contact them at:
NC-GAP Analysis Project
Dept. of Zoology, NCSU
Campus Box 7617
Raleigh, NC 27695-7617
(919) 513-2853
a target="_blank">www.basic.ncsu.edu/ncgap